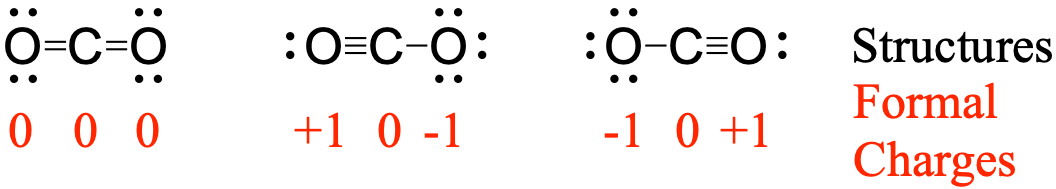
Chromium is a transition element and should lose its s electrons and then its d electrons when forming a cation. Zinc is a member of group 12, so it should have a charge of 2+, and thus loses only the two electrons in its s orbital. , Electron affinities from endothermic negative ion charge transfer reactions. For the p-block elements, electrons are removed from the p orbitals and then from the s orbital. For the transition metals, electrons are removed from the s orbital first and then from the d orbital. The reactive oxygen ion superoxide is particularly important as the product of the one-electron reduction of dioxygen O2, which occurs widely in nature. 1 The systematic name of the anion is dioxide (1). In terms of the elementary charge e, what is the total charge of. Next, remove electrons from the highest energy orbital. In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula O 2. Question: A particular ion of oxygen is composed of 8 protons, 10 neutrons, and 7 electrons. Write the electron configurations of these cations.įirst, write the electron configuration for the neutral atoms: Two of these essential trace elements, chromium and zinc, are required as Cr 3 + and Zn 2 +. They are called “essential” because they are required for healthy bodily functions, “trace” because they are required only in small amounts, and “elements” in spite of the fact that they are really ions. There are at least 14 elements categorized as “essential trace elements” for the human body. Unfortunately, the relative nature of these suffixes mandates that the ion formula/ion name combinations of the polyatomic ions must simply be memorized.\): Determining the Electronic Structures of Cations Additionally, both the nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions do not share a common suffix. Using a simple, general trend for the ionic charge for elements on the Periodic Table, in this video we find the ionic charge for Oxygen (O). While both the nitrate ion and the sulfate ion share an " -ate" suffix, the former contains three oxygens, but the latter contains four. However, the sulfate ion is symbolized as SO 4 −2. With Al 3 + and O 2, note that neither charge is a perfect multiple of the other. Valence electrons can be calculated by locating the position. We need three F ions to balance the charge on the Al 3 + ion, so the proper ionic formula is AlF 3. The formal charges can be calculated using the formula given below: The formal charge of an atom valence electrons of an atom non-bonding electrons (bonding electrons) The valence electrons (V.E) of an atom are the total number of electrons present in its valence shell. 
For example, the nitrate ion, which is symbolized as NO 3 −1, has one more oxygen than the nitrite ion, which is symbolized as NO 2 −1. We need two Cl ions to balance the charge on one Ca 2 + ion, so the proper ionic formula is CaCl 2. Note that all of the polyatomic ions whose names end in " -ate" contain one more oxygen than those polyatomic anions whose names end in " -ite." Unfortunately, these suffixes only indicate the relative number of oxygens that are contained within the polyatomic ions. Additionally, these suffixes also indicate the relative number of oxygens that are contained within the polyatomic ions. Therefore, " -ate" and " -ite" suffixes are employed, in order to denote that the corresponding polyatomic ions are part of a series. A single suffix, "-ide," is insufficient for distinguishing the names of the anions in a related polyatomic series. The remaining polyatomic anions, which all contain oxygen, in combination with another non-metal, exist as part of a series in which the number of oxygens within the polyatomic unit can vary. However, only two of these, the hydroxide ion and the cyanide ion, are named using the "-ide" suffix that is typically indicative of negatively-charged particles. There are a couple other possibilities which you may run into when studying free radical reactions and such. The remaining polyatomic ions are all negatively-charged and, therefore, are classified as anions. This is where a novel oxygen-ion battery technology comes into play, representing a vital component in the transition to cleaner energy sources. When oxygen bonds we have found it to either have a formal charge of 0 (2 bonds and 2 lone pairs), +1 (3 bonds and 1 lone pair), and -1 (1 bond and 3 lone pairs). Portable oxygen concentrators onboard the aircraft. The opposite charges of cations and anions exert a moderately strong mutual attraction that keeps the atoms in close proximity forming an ionic bond. The ionic form of selenium (Se), for example, is typically written Se 2. This polyatomic ion contains one nitrogen and four hydrogens that collectively bear a +1 charge. The items listed below will be accepted as Checked Baggage for a 75 each way charge. An anion that has accepted two electrons has a net charge of 2. 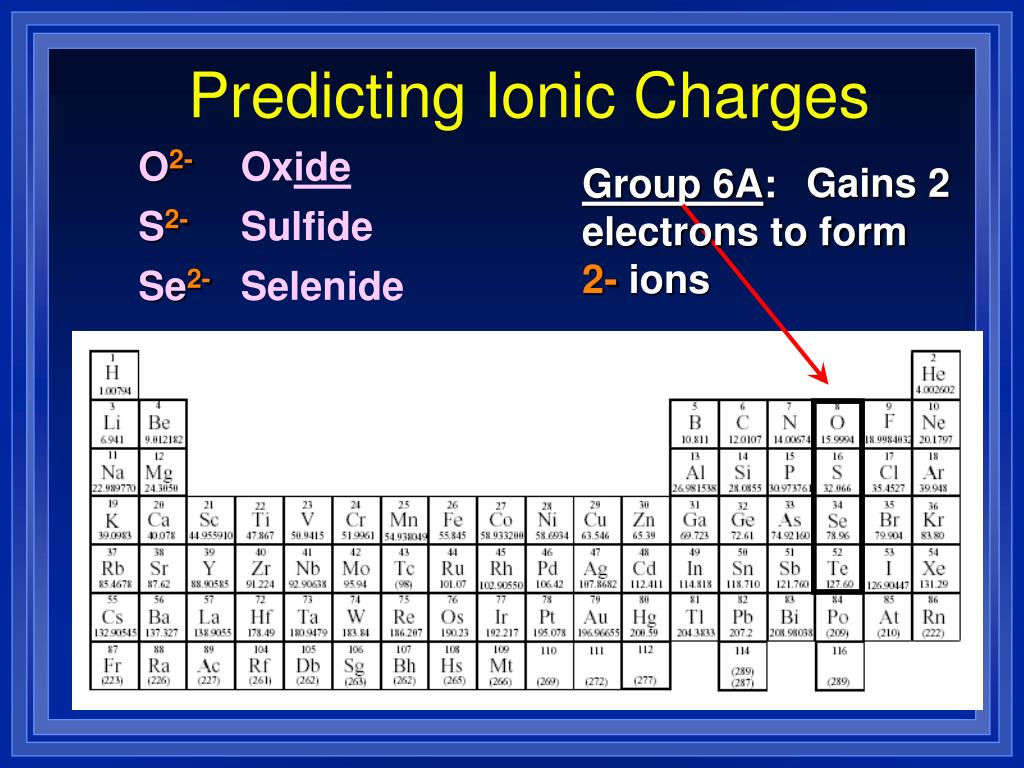
Note that only one polyatomic ion in this Table, the ammonium ion (NH 4 +), is a cation. \): Ion Names and Ion Formulas of Common Polyatomic Ions Ion Name
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
